Take the quiz and find out.
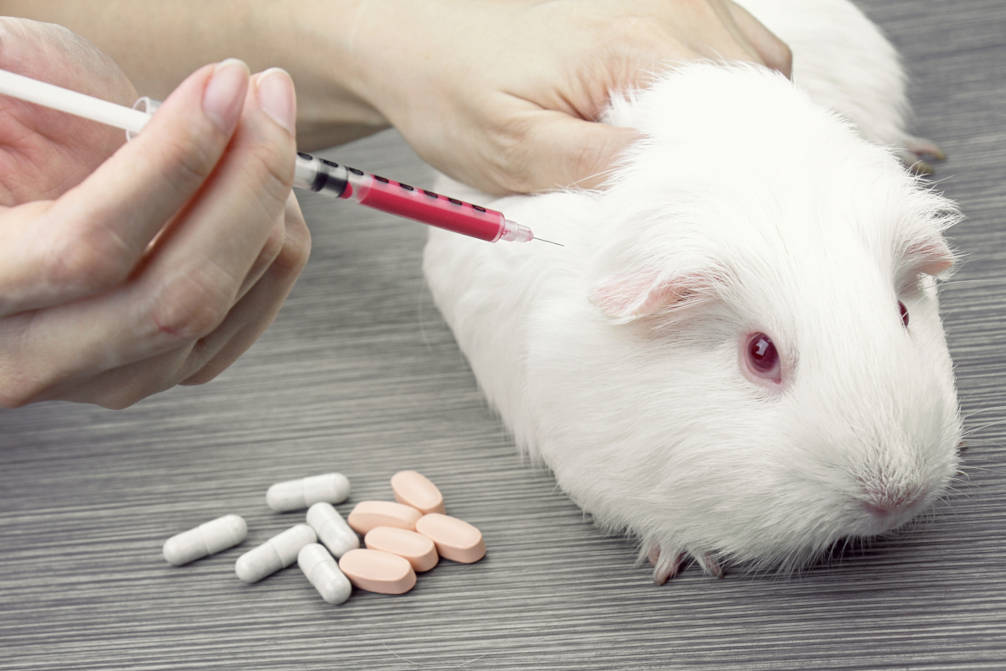
- How many animals are experimented on each year in SA, and which species?
- How much taxpayers’ money is spent on animal experiments in SA?
- How many animal experiments lead on to clinical trials?
- What kinds of research are being done?
- What sorts of procedures are carried out on animals? What level of suffering is involved?
- What happens to the animals at the end of experiments? If cats and dogs were rehomed from labs, as in NSW, how many animals would be involved?
- How often do ethics committees reject research proposals, or require serious amendments?
- To what extent are the 3Rs (Replacement, Reduction, Refinement) being implemented?
Chances are you scored 0/8, like just about everyone else. That’s because in SA there is a total lack of transparency regarding animal use for research. At least Victoria and NSW publish statistics each year, albeit belatedly. See:
Prior to the 2022 state election, the Labor party answered “yes” to the following question:
Will your party follow the lead of other states and publish an annual report of the number of animals used in research and teaching, the purposes for which they were used, and the fate of the animals?
So far nothing has happened, we still have the same secrecy surrounding animal experiments.
Statistics are only part of the story – they don’t answer many of our quiz questions. We need to be told more because animal experiments are being carried out in our name, with taxpayers’ money and purportedly for the public benefit. Clearly animal research is a public policy issue, and transparency by governments is essential to allow us as citizens to be meaningfully involved in debating such issues. We need to know much more about what’s going on and how decisions are made.
The approval process for research
Before a research project can begin, it must be approved by the Animal Ethics Committee (AEC) set up in each institution. The AEC must evaluate the project in accordance with the Australian Code for the Care and Use of Animals for Scientific Purposes. The Code sets out general principles, such as researchers must show respect for animals by:
- using animals only when justified
- supporting the wellbeing of the animals involved
- avoiding or minimising harm
- applying Replacement, Reduction and Refinement (3Rs) at all stages of animal use and care.
- Researchers must demonstrate that the project has scientific or educational merit, and that the use of animals is essential to achieve the stated aims and suitable alternatives to the use of animals are not available.
In fact, the Code (1.18) requires that: “Methods that replace or partially replace the use of animals must be investigated, considered and, where applicable, implemented.”
These principles sound reassuring to the public, suggesting that research is so well regulated there is nothing to worry about. But the public is not allowed to know anything about how AEC deliberations are conducted and how difficult questions are resolved. Members of the AEC are required to sign a confidentiality agreement, so they cannot discuss their work with anyone outside the committee, even if they have serious misgivings (1). The result is that the public cannot scrutinise the approval process, but is expected to endorse it on trust.
How could transparency be improved?
In the UK, and in EU member states as well, details of all experiments that are approved are published on the government website. This is done in a timely manner, so that by mid-year experiments approved in the first quarter are already available — see an example here. These reports are referred to as Non-Technical Summaries (NTS), and must be written in a way that is accessible to the general public.
An NTS has to include information relevant to the cost benefit analysis, such as:
- What are the aims of the project and what are its potential benefits?
- What outputs are anticipated and who will benefit from these outputs?
- What will be done to the animals?
- What are the expected impacts/adverse effects of these procedures and what is their expected severity?
- What will happen to the animals at the end of the experiment?
An NTS must address each of the 3Rs, that is, attempts to replace animals or reduce their number, and refine the procedure to minimise harm. With regard to Replacement, questions include: Why do you need to use animals? What non-animal alternatives have been considered? Why were they not suitable?
While publication of NTS would be an improvement on our current situation, it has potential limitations. A review of NTS in the UK and Germany, the two largest animal users in the EU, found that they were often of poor quality, for example, potential benefits exaggerated, adverse effects downplayed, and inadequate information on how the 3Rs were being applied.
On this issue, the authors observed (2: p.207):
“There was a tendency for NTS from both countries to have a relatively high proportion of generic statements providing assurances that the 3Rs were being applied, with no evidence provided to allow the reader to assess the veracity of this claim.”
The authors suggested the need for a “competent authority” to check the completeness and accuracy of the NTS in order for the public to be able to assess the project (2: p.209).
“We consistently found that the NTS were deficient in their description of what is actually being done to the animals and what they might experience as a result. This perhaps reflects a natural difficulty that researchers have in describing their experiments in simple terms and admitting that their work can cause harm to the animals. Competent authorities have a role to play in improving their guidance to researchers and ultimately ensuring that the NTS they produce is a fair representation of the project being authorised.”
Conclusion
Citizens in a democratic country have a right to know how government money is spent, and whether activities authorised by the government are consistent with community values. The secrecy surrounding animal research is unacceptable, especially since this is a very controversial issue. Therefore:
- South Australia must publish annual animal use statistics in a timely manner.
- The statistics should be collated in a nationally consistent way to enable comparisons between states.
- All states must publish Non-Techical Summaries of projects as soon as possible after they are approved.
- Ethics Committees must accept responsibiity for ensuring that the NTS are complete and accurate.
Action
Please email the Hon Susan Close and ask: what progress has been made towards the Labor commitment in 2022 to publish statistics of animals used in research? Send to officeofthedeputypremier@sa.gov.au
References
(1) Merkes, M. & Buttrose, R. (2019). Increasing the transparency of animal experimentation: an Australian perspective. In Herrmann, K. & Jayne, K. Animal experimentation: working towards a paradigm change. Brill, Leiden|Boston
(2) Taylor, K., Rego, L. & Weber, T. (2018). Recommendations to improve the EU Non-Techinical Summaries of animal experiments. ALTEX, 35 (2) 193-210

Humane Research Australia website has masses of up to date info.They say:Consider what medical research would take place without HRA scrutiny.HRA adds ethical context to inhumane experimentation.HRA advocates for animals used and killed.HRA holds government to account.Become a member and attend HRA zoom meetings —- next one Tuesday 15/8.They want to change their name to Animal Free Science Advocacy.Their mission is to end animal experimentation in our country.
Totally agree, Humane Research Australia is an excellent organisation and I urge everyone to join. However, HRA couldn’t answer the quiz questions either because the information in not publicly available and it should be. We need greater transparency.